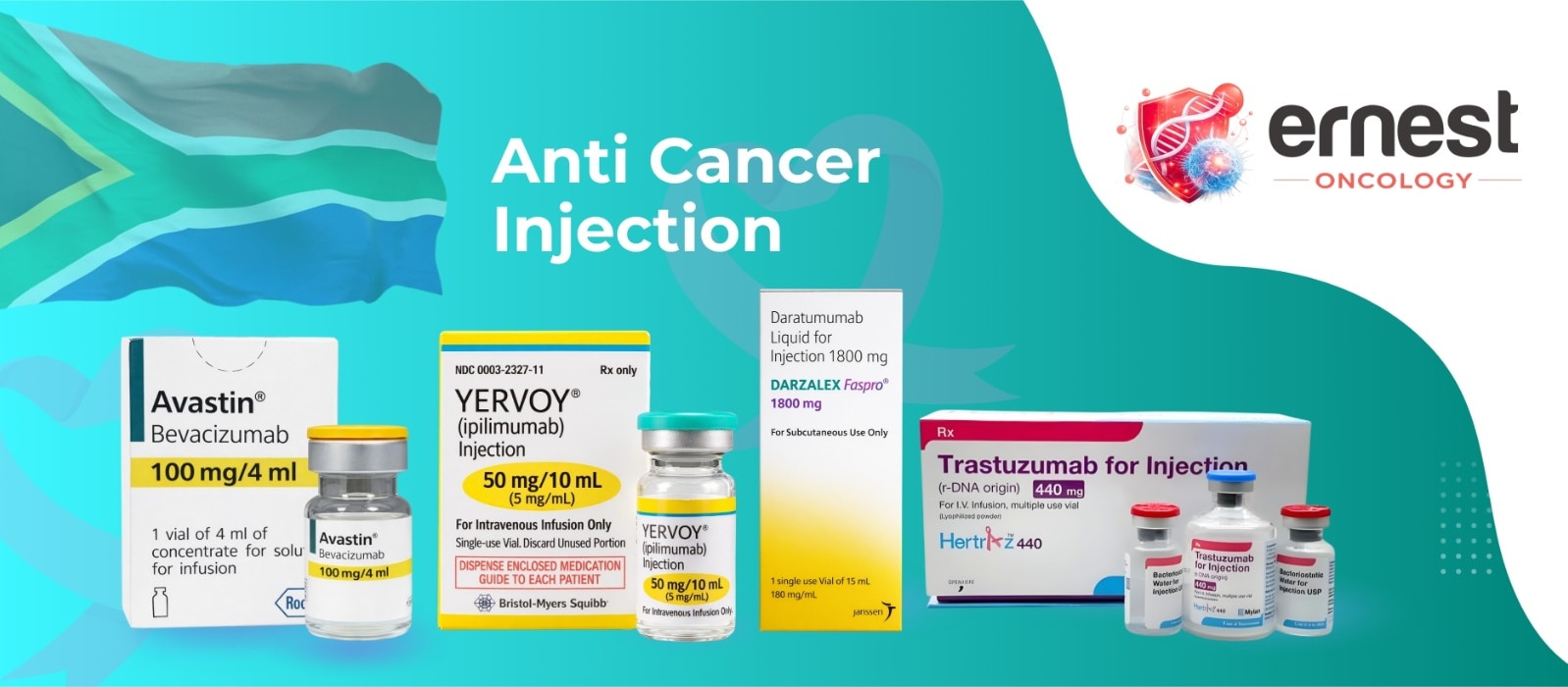
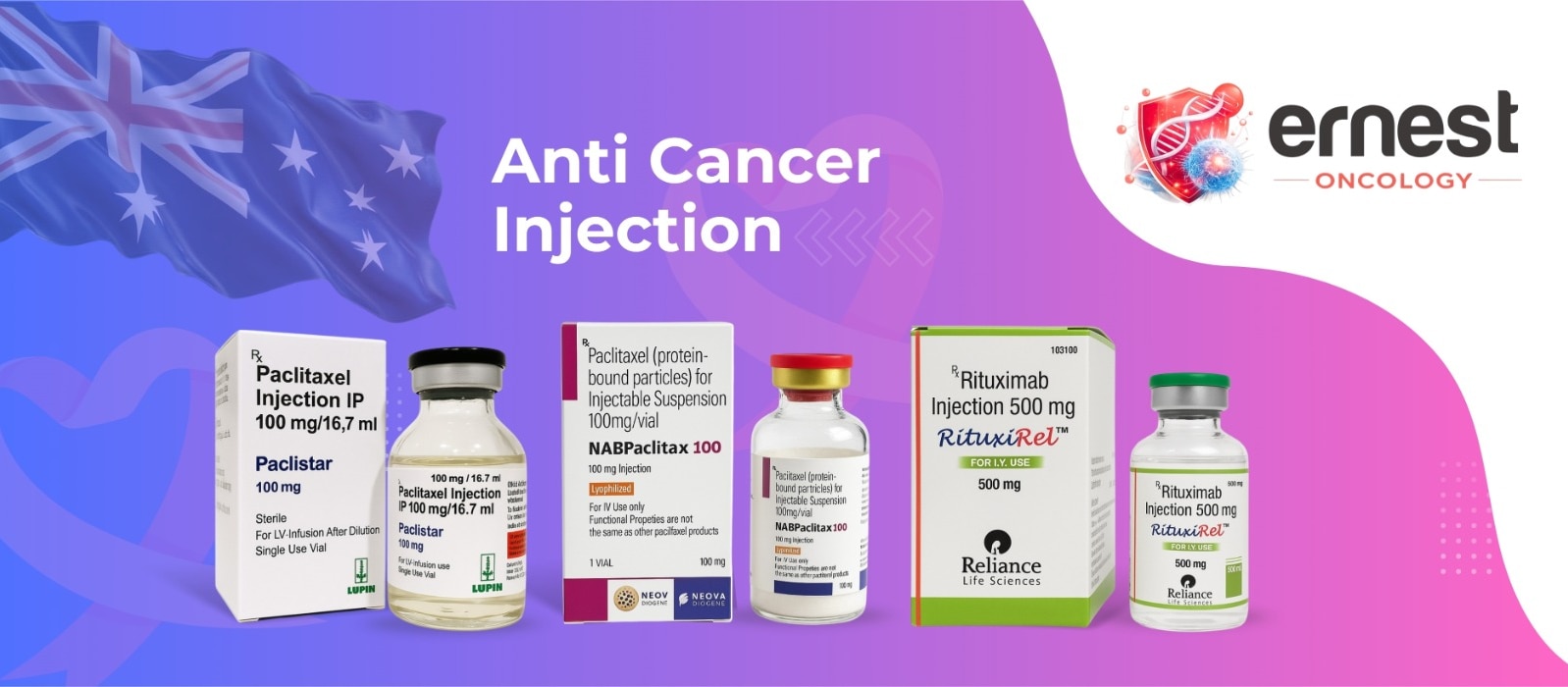
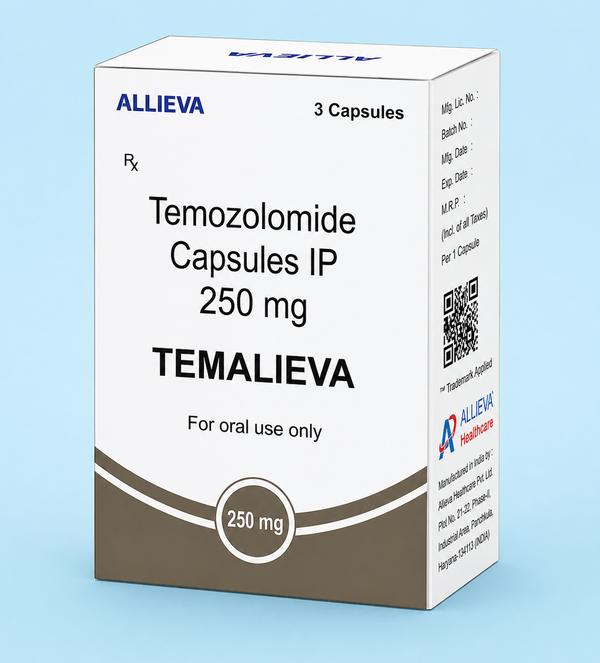
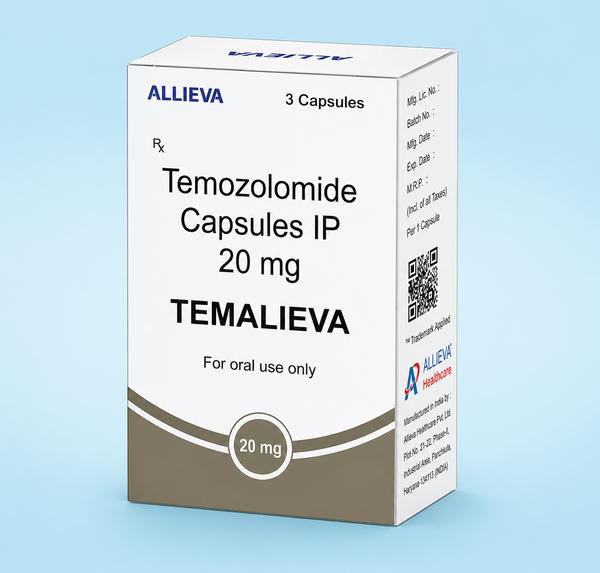
The Comprehensive Guide to Temalieva 250 mg Capsules In precision neuro-oncology and general cancer care, Temalieva 250 mg capsules (containing the active ingredient Temozolomide) stand as a critical therapeutic agent for treating aggressive brain tumors. As a premier pharmaceutical exporter from India, Ernest Oncology (a group of Ernest Pharmaceutical Pvt. Ltd.) ensures that this life-saving medication is accessible to hospitals, pharmacies, and healthcare systems in London, the UK, the USA, Australia, and South Africa with full regulatory compliance. Product SpecificationsFeatureDetailBrand NameTemalieva 250 (Temozolomide Capsules IP)Strength250 mgDosage FormOral Hard Gelatin CapsulePrimary IndicationGlioblastoma multiforme & anaplastic astrocytomaStorageStore below 25°C in a cool, dry place. Protect from moisture and light. Mechanism of Action: How It Works DNA Alkylation: Once ingested, Temozolomide readily crosses the blood-brain barrier. At physiological pH, it is converted into the active compound MTIC. Interfering with Replication: MTIC methylates DNA at specific locations, which causes DNA strand breaks and cross-linking. This process inhibits the multiplication and growth of rapidly dividing cancer cells, ultimately leading to programmed cell death (apoptosis). Therapeutic Indications & Administration Glioblastoma Multiforme (GBM): Used for newly diagnosed GBM patients in combination with focal radiation therapy, followed by maintenance therapy. Refractory Anaplastic Astrocytoma: Treatment for patients experiencing tumor recurrence or progression after standard therapy. Administration Guidelines Dosing Schedule: Dosing is calculated based on the patient's body surface area (BSA). Ingestion: Swallow the capsules whole with a full glass of water. Do not open, crush, or chew the capsule contents to prevent exposure to the active drug. Minimizing Side Effects: To reduce the incidence of nausea and vomiting, patients are often advised to take the medication on an empty stomach or at bedtime. Clinical Side Effects & Safety Profile Common Side Effects: Nausea, vomiting, headache, fatigue, constipation, and loss of appetite. Prophylactic antiemetics are frequently prescribed. Myelosuppression: The most serious clinical risk is a decrease in bone marrow function, which leads to reduced white blood cell and platelet counts. This increases susceptibility to infections and bruising or bleeding. Liver Function: Patients should undergo regular liver function tests (LFTs) due to the potential for hepatotoxicity. Contraindications: It is contraindicated during pregnancy and breastfeeding due to the risk of fetal harm. Male patients must use effective contraception during treatment and for at least 3 months afterward. Why Source from Ernest Oncology? United Kingdom (London): Providing high-quality, cost-effective generic alternatives for private and public healthcare systems. USA: Delivering bulk quantities that comply with international documentation and safety standards. Australia: Facilitating secure access through compliant supply channels such as the “Special Access Scheme“ (SAS). South Africa: Bridging critical gaps in the oncology market with affordable, high-tier pharmaceutical exports from India. 📞 Contact Ernest Oncology — Pharmaceutical Exporter from India [GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.] 📦 Business Type: Exporter | Bulk Supplier | Distributor 🌐 Websites: www.oncologymedicinesupplier.com www.ernestpharmaceuticals.com www.ernestvision.com www.ernestimpex.com 📧 Email: exports@ernestpharmaceuticals.com 📲 WhatsApp: +91 93599 02383 🔗 Direct Link: https://wa.me/919359902383

About ERNEST ONCOLOGY (Group of Ernest Pharmaceutical Pvt Ltd)
The best Manufacturer in Nagpur
Products
Featured ProductsWhat We Do
Image Gallery
Pictures That Speak
Have any custom requirements?
Updates
Read What’s Latest
×
![]()