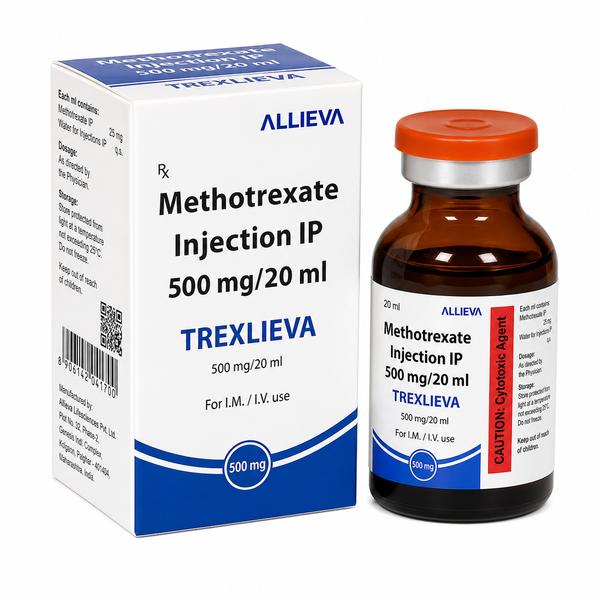
TREXLIEVA 500 MG Injection (Methotrexate): High-Strength Antimetabolite for Intensive Oncology Care TREXLIEVA 500 MG Injection is a high-concentration antineoplastic medication featuring the active pharmaceutical ingredient Methotrexate. As a potent folate antagonist, it is a critical component of high-dose chemotherapy regimens used to treat aggressive malignancies. Manufactured under rigorous GMP quality standards, TREXLIEVA 500 MG is engineered to deliver the intensive therapeutic intervention required for systemic cancer management across the USA, UK, Europe, UAE, and Asia. Therapeutic Indications and Global Clinical Usage Acute Lymphocytic Leukemia (ALL): Often used in intensification and maintenance phases to eliminate residual cancer cells. Osteosarcoma: A primary agent in high-dose regimens for malignant bone tumors, typically followed by leucovorin calcium rescue. Non-Hodgkin’s Lymphoma: Employed for systemic treatment and the prevention of central nervous system (CNS) involvement. Gestational Trophoblastic Neoplasia: Used in the treatment of choriocarcinoma and related trophoblastic diseases. Advanced Breast Cancer: Integrated into combination cycles for patients with metastatic disease Mechanism of Action: How TREXLIEVA 500 MG Works Enzyme Targeting: TREXLIEVA inhibits the enzyme dihydrofolate reductase (DHFR) with extreme precision. Folate Depletion: By blocking DHFR, the drug prevents the conversion of folic acid into its active form, tetrahydrofolate. DNA Synthesis Arrest: Tetrahydrofolate is a critical co-factor needed to create thymidylate and purines—the building blocks of DNA and RNA. S-Phase Specificity: The drug primarily affects cells in the S-phase of the cell cycle (when they are actively making new DNA), making it highly effective against rapidly dividing malignant cells. Cytotoxic Outcome: Without the ability to synthesize DNA, the cancer cell cannot divide and eventually undergoes programmed cell death. Professional Administration and Safety Protocols Route: Administered via Intravenous (IV) Infusion, Intramuscular (IM), or Intrathecal injection (in specific settings). High-Dose “Rescue“ Protocol: When using the 500 MG strength, administration of Leucovorin Calcium (Folinic Acid) is mandatory at specific intervals to protect healthy cells from lethal folate deficiency. Alkaline Diuresis: Patients receiving high doses must be aggressively hydrated and administered sodium bicarbonate to keep urine pH alkaline, preventing the drug from precipitating in the kidneys. Clinical Oversight: Must be administered by specialized oncology teams in settings equipped for real-time monitoring of serum methotrexate levels. Safety Profile and Clinical Monitoring Hematological Vigilance: Frequent Complete Blood Counts (CBC) are required to monitor for significant bone marrow suppression (neutropenia and thrombocytopenia). Nephrotoxicity Monitoring: Constant checks of serum creatinine and urine output are essential to prevent renal failure. Hepatotoxicity: Periodic liver function tests (LFTs) to monitor for treatment-induced hepatic strain. Mucositis Monitoring: Vigilance for severe inflammation of the mucous membranes, a common side effect of high-dose folate inhibition. Storage and Global Handling Instructions Storage: Store at controlled room temperature (20°C to 25°C) and protect the vial from light. Handling: As a hazardous cytotoxic agent, TREXLIEVA 500 MG must be prepared using specialized Personal Protective Equipment (PPE) and disposed of according to international biohazard protocols. Why Source TREXLIEVA 500 MG from Ernest Oncology? 100% Genuine Medication: Sourced directly from GMP-certified channels, ensuring every vial is authentic with an optimal shelf life. Global Distribution Network: Secure and rapid shipping to the USA, UK, UAE, Australia, Singapore, South Africa, Europe, Middle East, Africa, and Asia. Logistics Precision: Our team manages pharmaceutical exports with expertise, utilizing secure packaging and efficient transit routes to maintain product stability. Regulatory & Documentation Support: We handle all necessary export paperwork, including Certificates of Analysis (CoA) and permits, for seamless customs clearance. Bulk Supplier Advantage: As a dedicated distributor, we offer competitive wholesale rates to support oncology hospitals, research clinics, and government health departments. 📞 Contact Ernest Oncology — Pharmaceutical Exporter from India [GROUP OF ERNEST PHARMACEUTICAL PVT. LTD.] 🌐 Websites: www.oncologymedicinesupplier.com www.ernestpharmaceuticals.com www.ernestimpex.com www.ernestvision.com 📧 Email: [exports@ernestpharmaceuticals.com] 📦 Business Type: Exporter | Bulk Supplier | Distributor 📲 WhatsApp: +91 93599 02383 🔗 https://wa.me/919359902383